【JACS】中科院谭蔚泓院士、张鹏晖|协同免疫疗法新突破:“圆环”状双靶向核酸载体实现100%小鼠肿瘤长期生存
文章标题: Dual-Targeting Multivalent Aptamer-Drug Hybrids for Synergistic Cancer Immunotherapy
通讯作者: Penghui Zhang, Weihong Tan
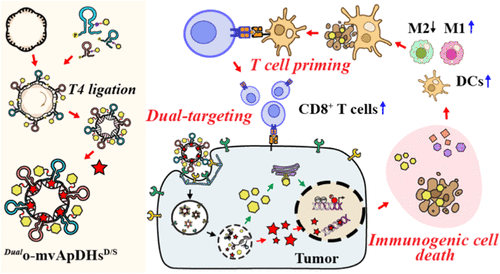
文章概要
引言
靶向药物偶联物在癌症治疗中已展现出巨大潜力,但传统的载体通常仅依赖单一抗原靶向和单一载药模式,这在面对高度异质性的肿瘤时往往显得力不从心,极易导致抗原逃逸和耐药性。为了克服这些局限,研究团队开发了一种创新的圆形双靶向多价适体-药物杂交体。该平台巧妙地利用了DNA纳米技术的编程特性,通过双重靶向机制来增强肿瘤的选择性。这项研究的核心思路在于将化疗诱导的免疫原性细胞死亡与先天免疫通路的精准激活相结合,通过协同作用彻底重塑免疫抑制性的肿瘤微环境,为下一代精密靶向药物的设计提供了全新的范式。
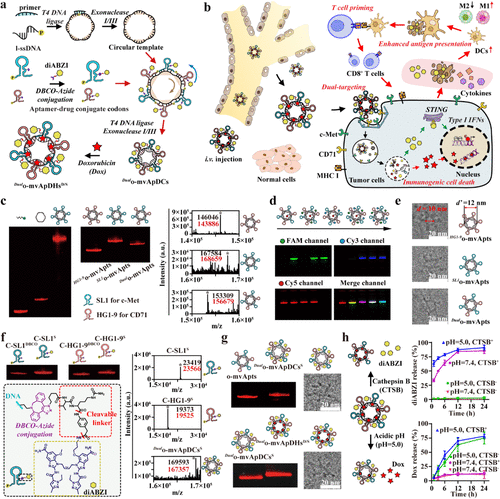
Figure 1. Circular dual-targeting multivalent aptamers for enhanced tumor accumulation. (a) Programmable construction of dual-targeting multivalent aptamer-drug hybrids. A circular single-stranded DNA template was generated by primer-guided circularization, T4 ligation and exonuclease purification. Sequence-defined aptamer or aptamer-drug conjugate codons were hybridized and ligated on the template. Doxorubicin intercalation into G-C pairs of the circular duplex yielded homogeneous _Dual_o-mvApDHsD/S with defined valence, flare-like architecture and high drug-loading capacity. (b) _Dual_o-mvApDHsD/S enabled synergistic antitumor immunity. Following intravenous injection, constructs accumulated in tumors via dual targeting of c-Met and CD71 and entered cells through multireceptor-mediated endocytosis. Intracellular Dox induced genotoxic stress and immunogenic cell death, whereas diABZI activated cGAS-STING to promote type I IFN signaling. The resulting DAMP- and STING agonist-rich debris was taken up by antigen-presenting cells, enhancing antigen presentation and cytotoxic T-cell priming. (c) Gel electrophoresis of circular multivalent aptamers (o-mvApts) incorporating SL1 (c-Met), HG1–9 (CD71) or both, with corresponding mass spectra. (d) Synthesis of o-mvApts encoding dye-labeled aptamer codons (FAM, Cy3, Cy5). Signals from individual and merged channels confirmed colocalization of all dyes within the same structure. (e) Representative cryo-EM images of _Dual_o-mvApts. The internal diameter of the structures (d’) was estimated at 12 nm, compared with the measured diameter (d) of 10 nm. (f) Characterization of aptamer-drug conjugate (ApDC) codons by gel electrophoresis and mass spectrometry. The inset shows the chemical structure of the diABZI conjugate bearing a cathepsin B-cleavable linker. The detected molecular masses are marked in black, and the corresponding calculated values are shown in red. (g) Characterization of _Dual_o-mvApDHsD/S by gel electrophoresis and cryo-EM images. (h) Controlled release of diABZI and Dox in the presence of cathepsin B or under acidic conditions. Data in panel h are presented as the mean ± standard deviation (s.d.).
主要实验及结论
研究人员首先通过序列引导的杂化与酶促木素聚合技术,成功构建了具有Flare(火焰状)结构的单链环状DNA模板。这种独特的环状拓扑结构彻底消除了暴露的末端,使其在血清中表现出极强的抗降解能力。在该载体上,团队同时装载了针对c-Met和CD71受体的双重适体,并协同递送化疗药物阿霉素(Dox) 和STING激动剂diABZI。实验证明,这种多价载体能够通过异核多受体介导的胞吞作用,显著提升肿瘤细胞对药物的摄取效率。在细胞内部,阿霉素引发强烈的基因毒性压力,诱导肿瘤细胞发生免疫原性细胞死亡,释放出大量的损伤相关分子模式;与此同时,释放出的diABZI激活了cGAS-STING信号通路,大幅增强了I型干扰素的反应。
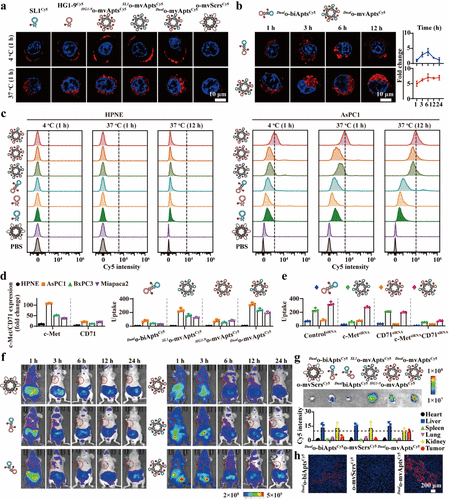
Figure 2. Targeted recognition and enhanced uptake of Dualo-mvApts. (a) Uptake of Cy5-labeled probes (each containing 100 nM Cy5) in AsPC1 cells at 4 or 37 °C over time. (b) Uptake kinetics of _Dual_o-biAptsCy5 and _Dual_o-mvAptsCy5 (each containing 100 nM Cy5) in AsPC1 cells. (c) Targeted recognition and uptake of o-mvAptsCy5 (each containing 100 nM aptamer) in HPNE cells and AsPC1 cells after incubation for indicated time. (d) Left panel: Flow cytometry analysis of c-Met and CD71 expression across different cell lines. Right panel: Cellular uptake of _Dual_o-biAptsCy5, _HG1–9_o-mvAptsCy5, _SL1_o-mvAptsCy5, and _Dual_o-mvAptsCy5 (n = 3) across different cell lines. (e) Cellular uptake of _Dual_o-biAptsCy5, _HG1–9_o-mvAptsCy5, SL1o-mvAptsCy5, and _Dual_o-mvAptsCy5 in AsPC1 cells following siRNA-mediated receptor knockdown (n = 3). (f) In vivo fluorescence imaging of Cy5-labeled probes (each containing 2 nmol Cy5) in AsPC1 tumor-bearing BALB/c nude mice (n = 3). (g) Ex vivo tumor fluorescence and quantification (n = 3). (h) Confocal imaging of tumor sections showing Cy5-labeled probes (red) and DAPI-stained nuclei (blue). Samples in panels g and h were collected 24 h after intravenous administration. Data in panels b, d, e, g are presented as the mean ± standard deviation (s.d.).
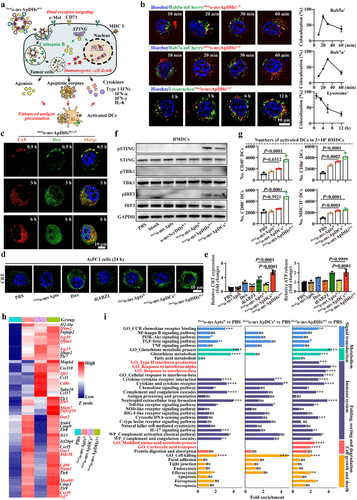
Figure 3. Dual-targeting multivalent aptamer-drug hybrids induce synergistic immunogenic cell death (ICD). (a) Schematic of synergistic ICD induced by _Dual_o-mvApDHsD/S. The hybrids entered tumor cells via dual targeting and heteromultireceptor, caveolae-dependent endocytosis. Intercalated Dox was released and diffused into the nucleus and mitochondria, whereas diABZI was cleaved by endogenous cathepsin B. The resulting STING agonists, DAMP-rich debris and cytokines activated antigen-presenting cells, enhancing antigen presentation and cytotoxic T-cell priming. (b) Intracellular trafficking of _Dual_o-mvApDHsCy5 in Rab5a-mCherry early endosomes, Rab7a-mCherry late endosomes, and Lysotracker-labeled lysosomes in AsPC1 cells. Colocalization was quantified using the Coloc 2 plugin in ImageJ (n = 10). (c) Monitoring Dox release from _Dual_o-mvApDHsD/Cy5 in AsPC1 cells. (d) Calreticulin (CRT) expression in AsPC1 cells by immunofluorescence. (e) Quantification of ICD by CRT exposure and ATP release in AsPC1 cells after 48 h of incubation with different formulations. (f) Activation of the cGAS-STING pathway in BMDCs following treatment. (g) Activation of BMDCs after exposure to cell debris. (h) Heatmap of differentially expressed genes (DEGs) from RNA-seq analysis of BMDCs treated with debris from AsPC1 cells versus naïve controls (n = 3). (i) KEGG analysis of major altered genes in mouse BMDCs treated with debris from AsPC1 cells versus naïve controls (n = 3). AsPC1 cells were treated with the indicated agents for 24 h, and the resulting debris was incubated with BMDCs for 12 h. Data in panels b, e, and g are presented as the mean ± standard deviation (s.d.). Statistical significance was assessed by one-way ANOVA with Tukey’s multiple-comparison test. P values: ns, not significant; *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
在针对胰腺癌(KPC)的小鼠模型实验中,这种双载药杂交体表现出了卓越的免疫重塑能力。单细胞转录组和TCR测序结果揭示,该疗法能够显著下调髓系抑制性细胞和调节性T细胞的比例,同时促进促炎型M1巨噬细胞的极化。这种环境的变化有效触发了CD8+ T细胞的增殖与功能维持,避免了T细胞进入终末耗竭状态。更令人振奋的是,当该杂交体与PD-1阻断剂联合使用时,协同效应达到了巅峰,不仅实现了所有实验小鼠超过90天的长期生存,甚至部分小鼠达到了肿瘤完全消退的效果,并产生了持久的免疫记忆,能够抵抗肿瘤的再次攻击。
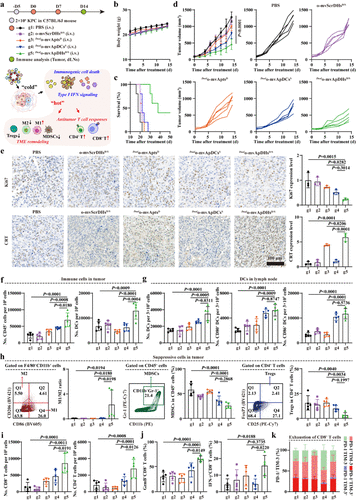
Figure 4. _Dual_o-mvApDHsD/S boost innate and adaptive antitumor immunity. (a) Schematic of intravenous administration of two doses of _Dual_o-mvApDHsD/S, _Dual_o-mvApDCsS, or _Dual_o-mvAptsD. The hybrids induced potent ICD, reshaping the immunosuppressive tumor microenvironment and coordinating innate and adaptive immunity. (b) Body weight changes and (c) Kaplan–Meier survival curves of mice following treatment (n = 5). (d) Tumor growth curves and individual tumor trajectories following treatment (n = 5). (e) Immunohistochemical staining and quantification of Ki67 and CRT in tumors on day 14, normalized to the PBS control (n = 3). (f) Numbers of tumor-infiltrating CD45+ immune cells and DCs (CD11c+MHC II+) in tumors on day 14 following treatment (n = 5). (g) Numbers of DCs and activated DCs (CD86+/CD80+) in draining lymph nodes (dLNs) on day 14 (n = 5). (h) Representative flow cytometry plots and frequencies of M1/M2 macrophages, MDSCs, and Tregs in tumors on day 14 (n = 5). (i) Numbers of CD8+ and CD4+ T cells in tumors on day 14 (n = 5). (j) Frequencies of GzmB+CD8+ and IFN-γ+CD8+ T cells in tumors on day 14 (n = 5). (k) Exhausted CD8+ T cells (PD-1+TIM-3+) in tumors on day 14 (n = 5). Data in panels b–k are presented as the mean ± standard deviation (s.d.). Statistical significance was assessed by one-way ANOVA with Tukey’s multiple-comparison test. P values: ns, not significant; *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
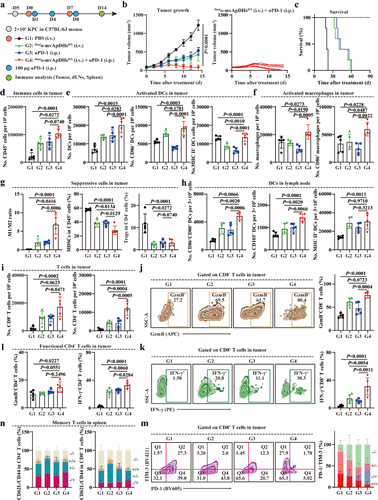
Figure 5. Combination of _Dual_o-mvApDHsD/S and ICB induces durable T cell responses. (a) Schematic illustration of _Dual_o-mvApDHsD/S+αPD-1 regimen in KPC-tumor bearing C57BL/6J mice. (b) Tumor growth curves following treatment and individual tumor trajectories following combination treatment with two doses of _Dual_o-mvApDHsD/S and three doses of αPD-1 (n = 5). (c) Kaplan–Meier survival analysis following treatment (n = 5). (d) Numbers of tumor-infiltrating CD45+ immune cells in tumors on day 14 following treatment (n = 5). (e) Numbers of DCs and activated DCs (CD86+/MHC II+) in tumors on day 14 (n = 5). (f) Numbers of macrophages and activated macrophages (CD86+) in tumors on day 14 (n = 5). (g) Frequencies of M1/M2 macrophages, MDSCs, and Tregs in tumors on day 14 (n = 5). (h) Activation of DCs (CD86+CD80+/ CD103+/MHC II+) in dLNs on day 14 (n = 5). (i) Numbers of CD8+ and CD4+ T cells in tumors on day 14 (n = 5). (j) Representative flow cytometry plots and percentage of GzmB+CD8+ T cells in tumors on day 14 (n = 5). (k) Representative flow cytometry plots and percentage of IFN-γ+CD8+ T cells in tumors on day 14 (n = 5). (l) Frequencies of GzmB+CD4+ and IFN-γ+CD4+ T cells in tumors on day 14 (n = 5). (m) Representative flow cytometry plots and percentage exhausted CD8+ T cells (PD-1+TIM-3+) in tumors on day 14 (n = 5). (n) Representative flow cytometry plots and percentage showing the subdivision of splenic CD4+ and CD8+ T cells into naïve (CD62L+CD44–), central memory (CD62L+CD44+), and effector memory (CD62L–CD44+) subsets on day 14 (n = 5). Data in panels b–n are presented as the mean ± standard deviation (s.d.). Statistical significance was assessed by one-way ANOVA with Tukey’s multiple-comparison test. P values: ns, not significant; *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
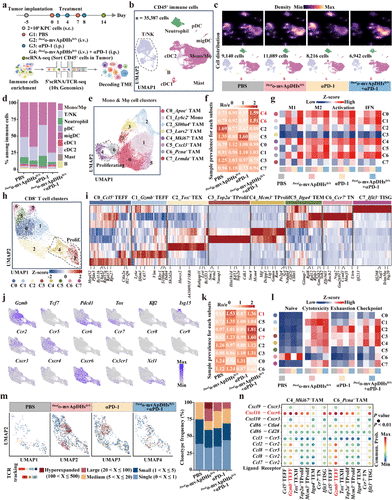
Figure 6. Single-cell transcriptomic landscape of tumor-infiltrating immune cells following combination therapy. (a) Schematic of the experimental design. Mice bearing KPC tumors were treated with the indicated formulations (n = 5). Tumors were collected on day 14, enzymatically digested to generate single-cell suspensions, and CD45+ immune cells were isolated for 5′ scRNA-seq with TCR enrichment. (b) Uniform manifold approximation and projection (UMAP) of major immune cell lineages, color-coded by cell type. (c) UMAP plots showing major immune lineages (bottom) and relative cell density (top) across the four groups; higher density is shown in bright magma. (d) Relative abundance of immune cell subsets across the four groups. (e) UMAP plots identifying subclusters within tumor-infiltrating monocytes/macrophages (Mono/Mφ), with color coding and cluster IDs indicated. (f) Heatmap showing the distribution of Mono/Mφ subclusters across groups, estimated by _R_o/e. (g) Heatmap of mean signature scores for each Mono/Mφ subcluster across groups. (h) UMAP plots identifying subclusters within the tumor-infiltrating CD8+ T cell compartment, with color coding and cluster IDs indicated. (i) Heatmap showing expression of differentially expressed genes (DEGs) across CD8+ T cell clusters; scaled expression is presented as z-scores. (j) Expression profiles of representative marker and chemokine receptor genes across the eight CD8+ T cell subsets. (k) Heatmap showing the distribution of tumor-infiltrating CD8+ T cell states across groups, estimated by _R_o/e. (l) Heatmap of mean signature scores for each CD8+ T cell subcluster across groups. (m) T cell clonotype frequencies across the experimental groups. (n) Bubble plots illustrating ligand–receptor pairs between Mki67+ TAM/Pcna+ TAM and the eight CD8+ T cell clusters, with color intensity indicating interaction strength.
总结及展望
该研究成功建立了一个模块化、可编程的多靶向多载药平台,证明了通过单一多功能分子同步实现肿瘤杀伤与免疫激活的可行性。这种圆形多价适体结构不仅在生物稳定性上表现优异,其模拟内源性环状DNA的物理特性也为其在复杂体内环境中的长循环和深层渗透提供了保障。未来,这一平台有望通过更换不同的适体和载荷,广泛应用于各类难治性实体瘤的精准治疗。这项成果标志着靶向药物偶联物从“单兵作战”正式跨入了“协同兵团”的新阶段,为开发更高效、更安全的下一代肿瘤免疫治疗药物奠定了坚实基础。