【JACS】苏州大学史海斌、程侠菊等|光控纳米疫苗能让放疗效果翻倍:原位捕获抗原与核酸的新策略
文章标题: Light-Mediated In Situ Synchronous Capture of Antigens and Nucleic Acids as Nanovaccines to Boost Tumor Radioimmunotherapy
通讯作者: Danyang Ji, Xiaju Cheng, Haibin Shi
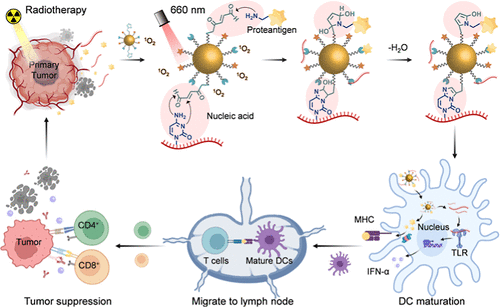
文章概要
引言
放射治疗是临床治疗实体瘤的基石,其诱导的免疫原性细胞死亡能够释放肿瘤抗原和损伤相关分子模式,理论上可以产生“原位疫苗”效应并激活全身免疫。然而,由于放疗释放的蛋白质抗原和核酸片段极易被体内的蛋白酶或核酸酶降解,且树突状细胞对这些成分的摄取效率有限,导致单纯放疗往往难以引发强效的远端效应。为了克服这一挑战,苏州大学的研究团队设计了一种创新的光中介策略,通过一种光响应纳米颗粒,实现在放疗现场对肿瘤抗原和核酸佐剂的同步捕获。这种方法不仅延长了免疫原性物质在肿瘤微环境中的滞留时间,还无需外源佐剂即可显著增强树突状细胞的成熟与抗原呈递,为恶性肿瘤的精准免疫治疗提供了新思路。
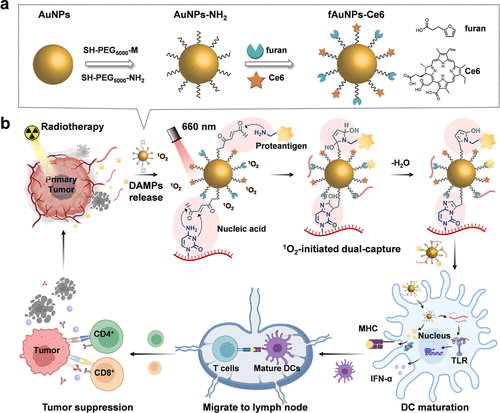
Scheme 1. Schematic Illustration of Light-Mediated Synchronous Capture of PAs and NA Adjuvants In Situ Generating Vaccines to Boost Tumor Radioimmunotherapy
主要实验及结论
研究人员开发了一种名为 fAuNPs-Ce6 的功能化金纳米颗粒,其表面修饰了光敏剂二氢卟吩e6和具有光化学活性的呋喃基团。实验证明,在660纳米激光照射下,光敏剂产生的单线态氧能触发呋喃基团与蛋白质中的氨基以及核酸中的碱基发生共价环化反应。体外实验数据表明,该系统捕获蛋白质和核酸的能力较对照组提升了约3倍,并能有效保护这些生物分子免受酶降解。在细胞层面,负载了抗原和核酸的纳米疫苗通过TLR7信号通路显著上调了树突状细胞表面CD80、CD86和MHC II的表达,并诱导了高水平的I型干扰素分泌。
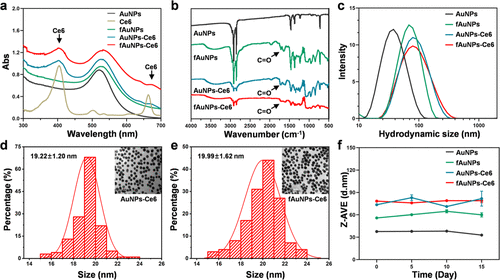
Figure 1. Chemical characterization of gold nanoparticles. UV–vis spectra (a), FT-IR spectra (b), and hydrodynamic sizes (c) of fAuNPs-Ce6 and other three control AuNPs in aqueous solution. The TEM images and particle sizes of AuNPs-Ce6 (d) and fAuNPs-Ce6 (e) nanoparticles (n > 100). (f) The hydrodynamic size change of all nanoparticles in 10% FBS-containing aqueous solutions over 15 days.
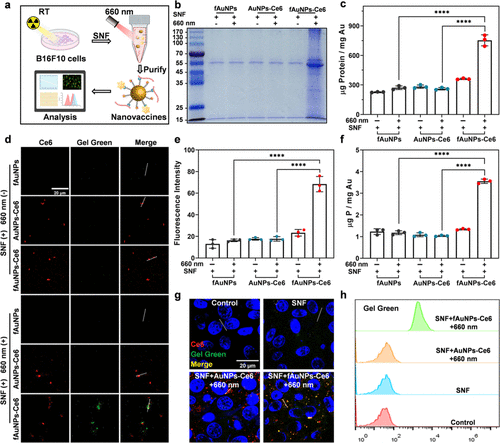
Figure 2. In vitro light-triggered PAs and NAs dual-capture by fAuNPs-Ce6 nanoparticles. (a) Schematic diagram of in vitro generating PAs/NAs-loaded nanovaccines. The capture of PAs by different AuNPs in the presence or absence of a 660 nm laser irradiation were analyzed by Coomassie blue staining of SDS-PAGE (b) and BCA assay (c,d) The amount of nucleic acids captured by different AuNPs in the presence or absence of a 660 nm laser irradiation were analyzed by confocal fluorescence microscope with GelGreen. (e) Quantitative analysis of nucleic acids captured by different AuNPs. (f) The concentration of P from NAs was determined by ICP-OES. The uptake of different AuNPs by DC2.4 cells was examined using confocal fluorescence microscope (g) and flow cytometry analysis (h) (SNF: the cell supernatant fluid of 8 Gy X-ray-irradiated B16F10 cells).
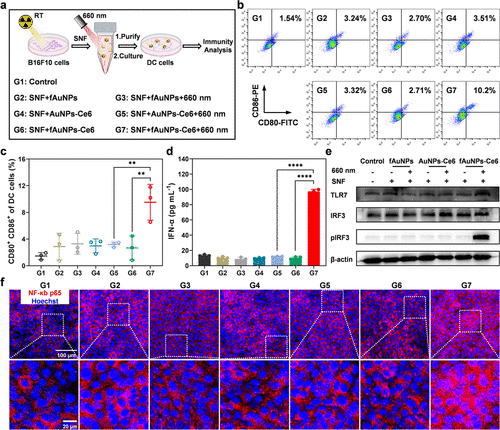
Figure 3. In vitro DC activation promoted by PAs/NAs-loaded fAuNPs-Ce6 nanovaccines. (a) Schematic diagram of in vitro generating PAs/NAs-loaded fAuNPs-Ce6 nanovaccines for DC activation. (b) Representative flow cytometry plots of the expression of CD80 and CD86 on BMDCs after different treatments. (c) Flow cytometric quantification of CD80+ CD86+ BMDCs after different treatments (n = 3). (d) IFN-α secretion by BMDCs after different treatments as indicated (n = 3). (e) Expression levels of TLR7, IRF3, and phosphorylated IRF3 proteins in DC2.4 cells after different treatments. (f) The nuclear translocation of NF-κB p65 (red) in DC2.4 cells after different treatments. Data are shown as mean ± SD. Statistical significance was assessed by ANOVA with Tukey’s post-test. p value: *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001 (SNF: the cell supernatant fluid of 8 Gy X-ray-irradiated B16F10 cells).
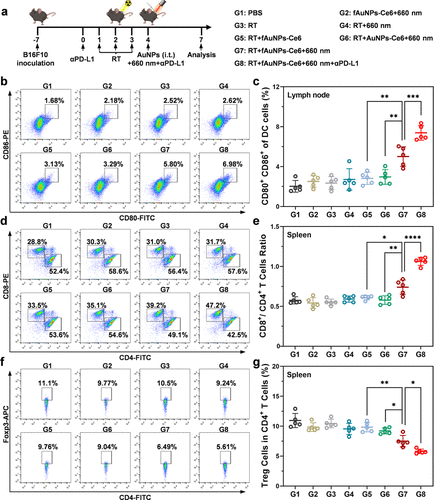
Figure 4. Immune response activation induced by fAuNPs-Ce6 nanoparticles in vivo. (a) Scheme for the establishment of B16F10 tumor-bearing mouse model and the treatment procedure. Representative flow cytometry plots (b) and relative quantitative analysis (c) of the expressions CD80 and CD86 in DCs of draining lymph nodes (n = 5). Representative flow cytometry plots (d) and relative quantitative analysis (e) of CD8+ T cells and CD4+ T cells (gated in CD3+) in spleen after different treatments (n = 5). Representative flow cytometry plots (f) and relative quantitative analysis (g) of Foxp3+ CD4+ Treg cells in spleen after different treatments (n = 5). Data are shown as mean ± SD. Statistical significance was assessed by ANOVA with Tukey’s post-test. p values: *p < 0.05, **p < 0.01, ***p < 0.001, and ****p < 0.0001.
在荷瘤小鼠模型中,研究团队系统评估了该策略的体内抗肿瘤效果。实验发现,fAuNPs-Ce6在瘤内注射后能高效富集于引流淋巴结。当联合放疗与PD-L1抑制剂时,该方案不仅彻底抑制了原发肿瘤的生长,还展现出强大的远端抑制作用,使未受照射的远端肿瘤体积缩减至对照组的数分之一。流式细胞术分析证实,这种综合疗法诱导了更高比例的CD8+效应T细胞浸润,并显著增加了体内记忆性T细胞的数量。更具临床意义的是,该策略在肿瘤再挑战实验中成功防止了肿瘤的复发,证明了其具有持久的免疫记忆功能。此外,通过对比仅捕获蛋白抗原的传统策略,本研究进一步确证了同步捕获核酸作为内源性佐剂对提升疫苗效力的不可替代性。
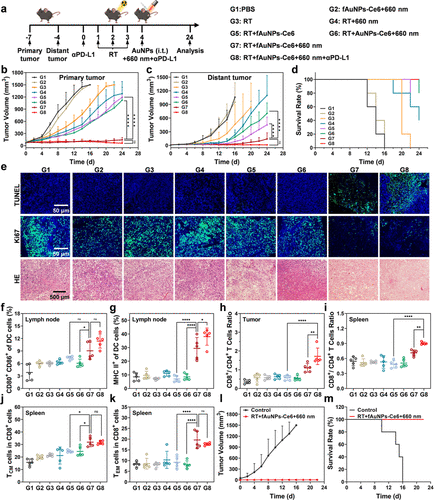
Figure 5. The therapeutic efficacy of tumor-bearing mice with various treatments. (a) Scheme for the construction of the bilateral B16F10-Luc tumor-bearing mice model and treatment procedure. The volumes of the primary (b) and distant tumors (c) of the mice with different treatments (n = 5). (d) Survival curves of the mice with different treatments (n = 5). (e) Staining of TUNEL, Ki67, and H&E in primary tumors of the mice with different treatments. Flow cytometric quantification of CD80+ CD86+ cells (f) and MHCII+ cells (g) of DCs in inguinal lymph nodes after different treatments (n = 5). Flow cytometric quantification of CD8+/CD4+ T cells in distant tumor (h) and spleen (i) of the mice after different treatments (n = 5). Flow cytometric quantification of central memory CD8+ T cells (j) and effector memory CD8+ T cells (k) in the spleen of mice after different treatments (n = 5). (l) The volumes of the recurrence tumors of mice with different treatments (n = 5). (m) Survival curves of the mice with different treatments (n = 5). Data are shown as mean ± SD. Statistical significance was assessed by ANOVA with Tukey’s post-test. p value: *p < 0.05, **p < 0.01, ***p < 0.001, and ****p < 0.0001. For survival analysis, the statistical differences on survival rate in each group were calculated by the Kaplan–Meier method.
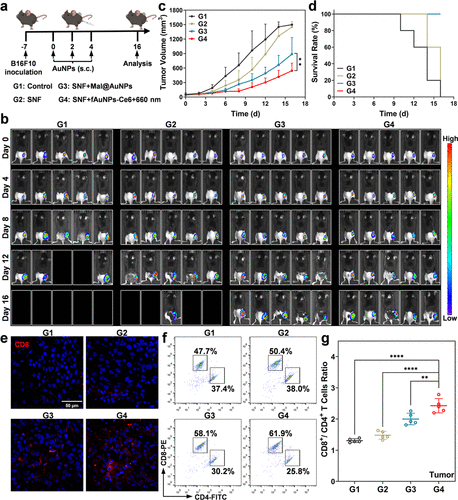
- Figure 6. Therapeutic efficacy of different nanovaccine strategies in vivo. (a) Schematic illustration of the administration strategy of therapeutic nanovaccines. (b) Bioluminescence imaging of the B16F10-Luc tumor-bearing mice with different treatments (n = 5). (c) The tumor volume changes for the mice with different treatments (n = 5). (d) Survival curves of the mice with different treatments (n = 5). (e) Immunofluorescent staining of CD8+ T cells in the tumors receiving different treatments. Representative flow cytometry plots (f) of CD8+ T cells and CD4+ T cells in tumors receiving different treatments and the relative quantitative analysis (g) (n = 5). Data are shown as mean ± SD. Statistical significance was assessed by ANOVA with Tukey’s post-test. p values: *p < 0.05, **p < 0.01, ***p < 0.001, and ****p < 0.0001. For survival analysis, the statistical differences on survival rate in each group were calculated by the Kaplan–Meier method (SNF: the cell supernatant fluid of 8 Gy X-ray-irradiated B16F10 cells).
总结及展望
这项研究成功展示了一种基于光化学反应的原位纳米疫苗构建平台,其核心优势在于能够同时锁定放疗产生的多种免疫成分。这种双重捕获策略极大改善了抗原递送的生物利用度,将放疗从一种局部物理治疗手段转化为强有力的全身免疫动员。由于该系统具有良好的生物安全性且不依赖复杂的体外制备流程,这种光控原位接种方法在未来的临床转化中具有广阔的前景,有望为那些对传统放疗或免疫检查点阻断不敏感的肿瘤患者带来更有效的治疗方案。