【JACS】北理工陈人杰、叶玉胜|锂电快充新突破!800次循环容量保持率超82%
文章标题: Probing Mesoscopic Solvation Dynamics via Comparable-Sized Nanomolecular Clusters
通讯作者: Yusheng Ye (叶玉胜), Renjie Chen (陈人杰)
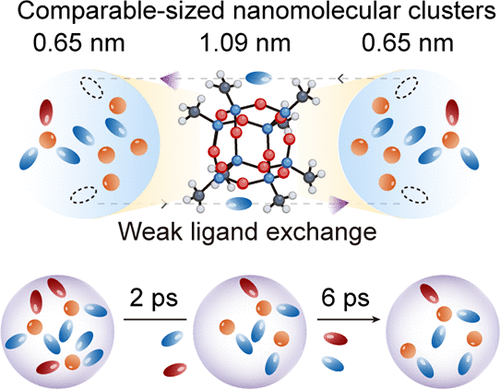
文章概要
引言
电解液的溶剂化动力学是决定生物、化学及电化学系统中质量传输过程的核心要素,尤其在锂电池领域,溶剂化结构直接影响着离子的迁移速率和界面反应动力学。长期以来,研究者们虽然意识到溶剂化壳层的重要性,但受限于观测手段,对于介观尺度下的 “壳层间”相互作用如何影响离子迁移仍知之甚少。传统的强溶剂化虽能促进盐的解离,却带来了巨大的脱溶剂化能垒,限制了电池的快充性能。为了破解这一难题,北京理工大学陈人杰教授、叶玉胜副教授团队在《美国化学会志》(JACS)上发表了最新研究成果。他们提出了一种创新的分子设计策略,通过引入尺寸与溶剂化壳层相匹配的纳米分子簇,实现了对介观溶剂化动力学的实时探测与精准调控,为开发高功率电解液开辟了全新路径。
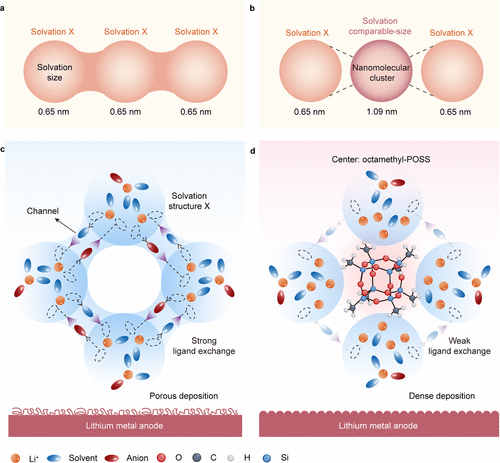
Figure 1. Proof-of-concept for intersolvation interactions mediated by octamethyl-POSS nanomolecular clusters with solvation-comparable sizes. a-b Schematic representations of intersolvation interactions in high concentration electrolytes, showing the system without (a) and with (b) octamethyl-POSS nanomolecular clusters. c-d Illustration of the ligand exchange mechanism, highlighting the absence (c) and presence (d) of octamethyl-POSS nanomolecular clusters and their effect on solvation dynamics.
主要实验及结论
研究团队选用性质稳定的八甲基多面体齐聚倍半硅氧烷(octamethyl-POSS) 作为介观探针。这种直径约为1.09纳米的分子簇与典型的锂离子溶剂化壳层尺寸相当,能够有效地嵌入并调节溶剂化网络。通过分子动力学模拟发现,在常规高浓度电解液中,锂离子被溶剂分子紧紧包裹,配体交换极其频繁且复杂。而加入POSS纳米簇后,锂离子的溶剂化环境发生了根本性重塑,锂离子在短短2皮秒内即可完成配体脱离,并在6皮秒内围绕POSS框架建立起全新的、弱配位的稳定构型。这种转变显著削弱了溶剂间的配体交换,降低了电解液的分解风险。
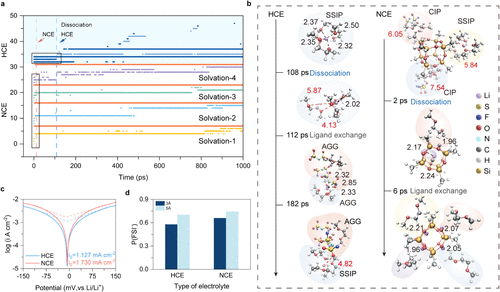
Figure 2. Molecular dynamics simulations reveal Li+transport mechanisms at 298 K. a Ligand residence times within 3 Å of Li+ in HCE (blue background) and NCE (white background), with red, dark, and light traces representing octamethyl-POSS, DME, and FSI–, respectively. Solid boxes highlight the dissociation events. b Time evolution of Li+ solvation dynamics in HCE (left) and NCE (right). c Tafel plots of Li|Li symmetric cells with different electrolytes under an applied potential of ±150 mV. d Probability of FSI– occurrence within the Li+ solvation shells (3 and 5 Å) at 298 K.
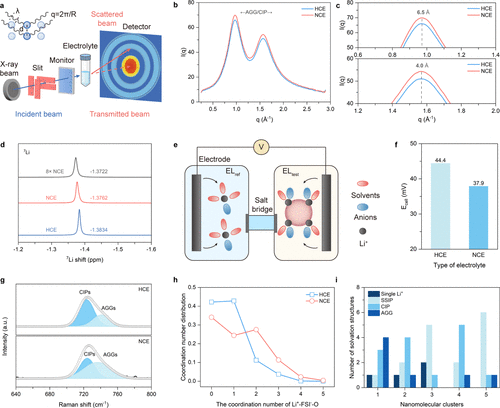
Figure 3. Electrolyte properties shaped by intersolvation interactions. a Schematic of the laboratory SAXS/WAXS setup. b-c WAXS patterns collected at 25 °C, shown over the full q range of 0.3–2.8 Å–1 (b) and in a magnified view highlighting structural differences (c). d 7Li NMR spectra of HCE, NCE, and concentrated 8× NCE (14.88 mmol), showing distinct Li+ environments. e Schematic illustrating the approach for extracting solvation energies. f Open-circuit voltages of HCE and NCE measured in symmetric Li|Li H-type cells. g Raman spectra comparing key coordination features in HCE and NCE. h Distribution of Li+-FSI–-O coordination numbers derived from simulation. i Solvation configuration distribution of octamethyl-POSS within 3 Å of a single Li+, revealing preferential coordination of Li+ with POSS-O.
在理化性质表征方面,7Li NMR谱图显示锂离子共振峰发生了明显的下移,证实了POSS纳米簇对锂离子的强烈吸引所产生的去屏蔽效应。通过电位测量进一步证实,新型纳米簇电解液具有更小的溶剂化自由能,这意味着锂离子在电极界面脱溶剂化变得更加轻而易举。拉曼光谱和模拟计算结果显示,POSS的加入将电解液结构从接触离子对占主导诱导转向了阴离子富集的聚集体结构。在外部电场的作用下,这种阴离子富集态进一步增强,促使在锂金属负极表面形成了一层更薄且富含无机物(如LiF和Li2O)的固态电解质界面膜(SEI)。
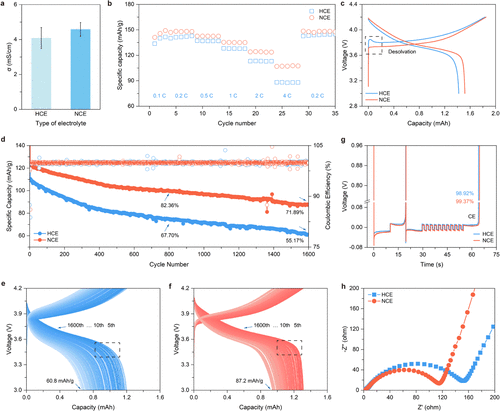
Figure 4. Electrochemical performance influenced by intersolvation clusters. a Ionic conductivities of different electrolytes. Error bars represent the mean ionic conductivity ± standard deviation (n = 5). b-c Rate capability (b) and first-cycle charge/discharge curves (c) of Li|NMC532 cells using HCE and NCE. d Long-term cycling of discharge capacity (filled symbols) and CE (open symbols) at 2 C within 3.0–4.2 V; upward and downward arrows indicate capacity after 800 and 1600 cycles, respectively. e-f Charge/discharge curves of HCE (e) and NCE (f) during 1600 cycles, sampled every 5 cycles from the fifth cycle. g CEs of HCE and NCE in Li|Cu cells (Aurbach method). h EIS spectra of fresh Li|NMC532 full cells with HCE and NCE.
电化学测试结果令人振奋。得益于优化的溶剂化结构和更快的离子迁移速率,该电解液将离子电导率提升至4.580 mS/cm。在Li|NMC532全电池测试中,使用该电解液的电池表现出极其优异的倍率性能和循环稳定性。在2 C的高倍率下循环800次后,容量保持率高达82.36%,远超传统高浓度电解液的67.70%。即使在更极端的1600次循环后,其容量保持率仍能维持在71.9%。此外,原位光学显微镜观察证实,这种电解液能有效抑制锂枝晶的生长,促进致密的纤维状锂沉积,确保了高电流密度下的安全性。
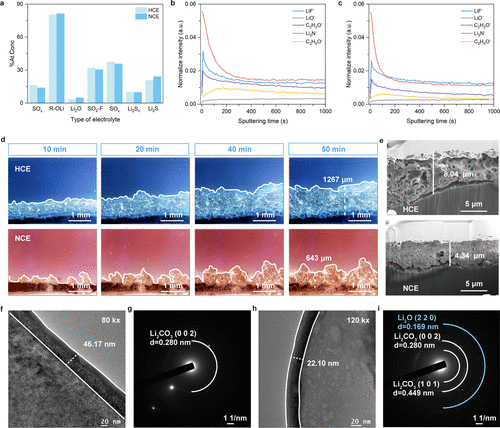
- Figure 5. Interfacial chemistry governed by the intersolvation interactions. a Statistical diagram of SEI composition extracted from O 1s and S 2p XPS spectra (0 s sputtering) of Li|NMC532 full cells after 10 cycles at 2 C in HCE and NCE. b-c TOF-SIMS depth profile curves of LiF–, LiO–, C2H2O–, Li3N–, and C2H5O– fragments in HCE (b) and NCE (c). d In-situ optical microscopy of Li|Li cells during deposition at 1 mA cm–2 (250×). Colors are inverted (blue, HCE; red, NCE;). e Cross-sectional FIB-SEM images (6000×) of Li anodes after 10 cycles at 2 C in Li|NMC532 cells. f–i Cryo-TEM analysis of SEI layers on Li0 deposits formed on Cu TEM grids (0.5 mA cm–2, 0.5 mAh cm–2). SEI morphologies in HCE and NCE at 80 and 120 k× (f, h), with thickness marked by dashed lines; corresponding SAED patterns (g, i) display crystalline phases.
总结及展望
本研究成功构建了一套基于纳米分子簇的介观溶剂化探测框架,揭示了壳层间相互作用对离子传输的关键影响。通过精细调控锂离子的配位环境,研究团队不仅实现了电解液本体电导率的提升,更通过工程化设计改善了界面脱溶剂化动力学。这种 “介观设计策略” 具有极强的普适性,不仅能显著提升锂金属电池的快充稳定性与循环寿命,还可推广至其他高性能二次电池及电化学系统中。这一成果为解决电动汽车和航空领域对高功率、长寿命能源存储技术的需求提供了重要的理论支撑和实践方案。