【JACS】中科院物理所胡勇胜利、容晓晖联合上海大学吕迎春、内蒙古大学徐淑银|能量密度突破268 Wh/kg!高钠P2型正极材料设计揭示钠离子电池新范式
文章标题: Rational Design of High-Na P2-Type Cathodes for Sodium-Ion Batteries: Unveiling Phase Formation Principles and Composition–Structure–Performance Correlations
通讯作者: Shuyin Xu, Xiaohui Rong, Yingchun Lyu, Yong-Sheng Hu
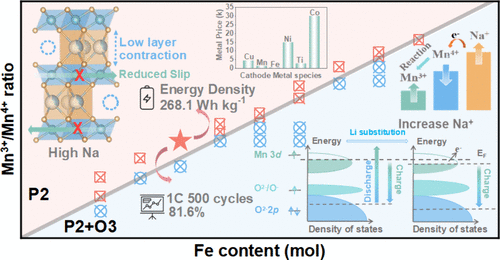
文章概要
引言
随着全球对于低成本、可持续能源存储方案的需求日益迫切,钠离子电池(SIBs) 凭借钠资源的广泛分布和极具竞争力的成本优势,已成为大规模储能领域中替代锂离子电池的关键技术。在众多正极材料中,P2型层状氧化物因其较宽的钠离子扩散通道和卓越的倍率性能而备受关注。然而,传统的P2型材料通常存在初始钠含量不足(通常x≈0.67)的问题,这不仅限制了电池的初始容量,还容易在深度脱钠过程中诱发不可逆的相变,导致循环寿命缩短。此外,此类材料在空气中的稳定性以及生产成本也是制约其商业化的瓶颈。为了解决这些挑战,本研究提出了一种基于高钠含量设计的全新策略,旨在通过优化过渡金属组分与锂离子掺杂,构建兼具高能量密度、长寿命和高稳定性的新型正极材料。
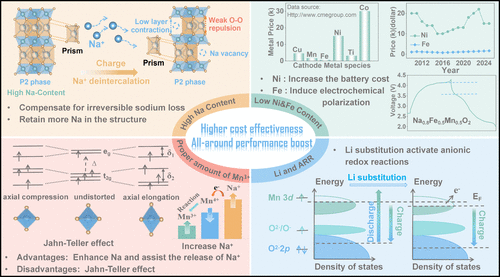
Figure 1. Four strategic approaches for engineering high-performance P2 phase layered cathode materials.
主要实验及结论
研究团队首先通过系统性的成分调节,深入探讨了钠-锂-铁-镍-锰-氧(Na–Li–Fe–Ni–Mn-O) 体系中的相形成规律。实验发现,在特定的铁含量下,增加三价锰离子的比例有助于稳定纯P2相,而较高的四价锰含量则容易导致P2和O3相的共生。基于这一发现,研究者利用线性回归分析建立了成分与性能之间的关联模型,并筛选出了最优配比方案:(简称N0.8LFNMO)。该材料采用了极低含量的昂贵镍元素,通过铁和锰的氧化还原贡献以及锂离子引发的阴离子氧化还原,实现了性能与成本的平衡。
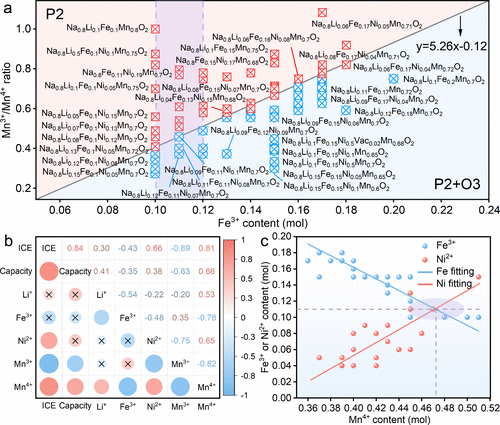
Figure 2. (a) Phase map for Na–Li–Fe–Ni–Mn-O series materials based on Fe content and Mn3+/Mn4+ ratio. (b) Correlation analysis between electrochemical performance and key factors. Red denotes a positive correlation, blue denotes a negative correlation, and a cross symbolizes a relatively weak correlation. (c) Data fitting relationship between Fe, Ni, and Mn4+.
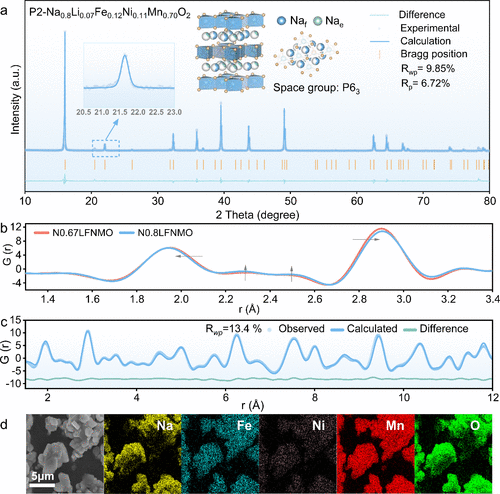
Figure 3. (a) Rietveld refinements of the X-ray diffraction patterns of N0.8LFNMO. (b) PDF profiles of N0.67LFNMO and N0.8LFNMO. (c) Refined PDF results of N0.8LFNMO. (d) SEM images and EDS mapping results of as-synthesized N0.8LFNMO.
结构表征结果显示,N0.8LFNMO具有清晰的六方P2型结构,且较高的初始钠含量显著提升了钠离子在晶格中的扩散动力学。原位XRD技术揭示了该材料在充放电过程中极佳的结构韧性,其晶格体积变化仅为1.5%,这一数值远低于同类材料,有效地抑制了过渡金属层的滑动和结构崩坏。在电化学性能方面,该正极在半电池中展现出125.8 mAh/g的可逆容量,并具有优异的倍率表现。当与硬碳负极匹配组成全电池时,系统实现了268.1 Wh/kg的超高能量密度,且在1C倍率下循环500次后容量保持率高达81.6%。此外,该材料展现出了卓越的空气稳定性,在湿度为60%的环境中暴露10天后,仍能保持结构完整并保留96.9%的初始容量,极大地降低了材料的存储与加工成本。
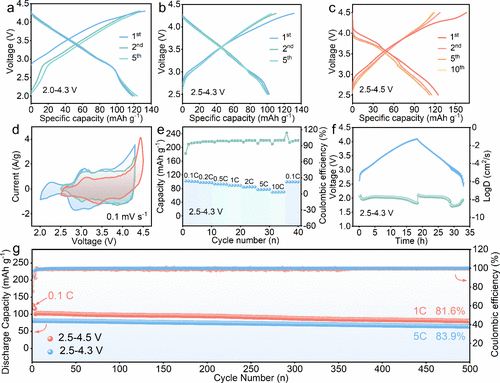
Figure 4. Electrochemical performance of N0.8LFNMO in half-cells. Charge/discharge curves of the N0.8LFNMO electrode in the voltage ranges of (a) 2.0–4.3, (b) 2.5–4.3, and (c) 2.5–4.5 V at 0.1 C rate. (d) Corresponding CV curves of the N0.8LFNMO electrode at 0.1 mV s–1. (e) Rate capability from 0.1 to 10 C. (f) GITT profiles of the N0.8LFNMO. (g) Cycling performance.
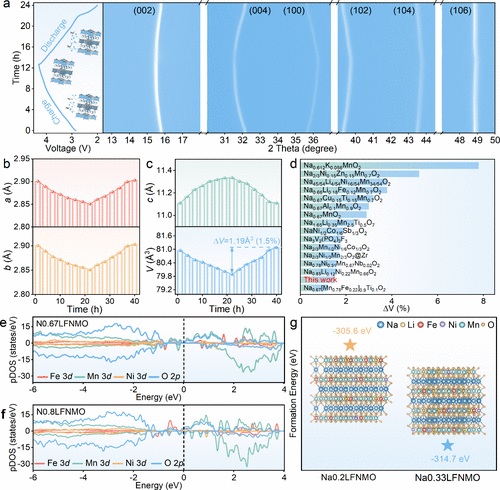
Figure 5. Structural evolution. (a) In situ XRD patterns of N0.8LFNMO during the first cycle. Corresponding (b) a and b axis parameters, (c) c axis and unit cell volume variations calculated from the in situ XRD results. (d) Comparison of the maximum cell volume changes in previously reported P2-type cathodes. Partial density of states (pDOS) of Fe 3d, Mn 3d, Ni 3d, and O 2p orbitals for (e) N0.67LFNMO and (f) N0.8LFNMO. (g) Formation energy of highly desodiated N0.2LFNMO and N0.33LFNMO.
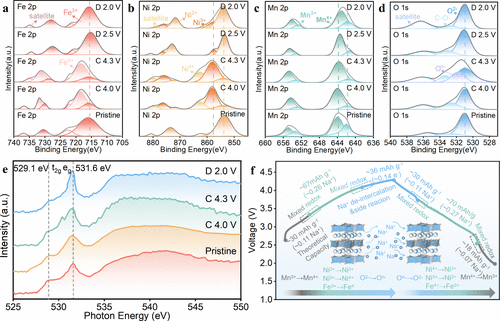
Figure 6. Charge compensation mechanism. Ex situ XPS spectra of (a) Fe 2p, (b) Ni 2p, (c) Mn 2p, and (d) O 1s orbital of N0.8LFNMO electrode charged at different states. (e) O K-edge s-XAS spectra of N0.8LFNMO electrodes collected at different charge and discharge states. (f) Schematic illustration of the charge compensation mechanism.
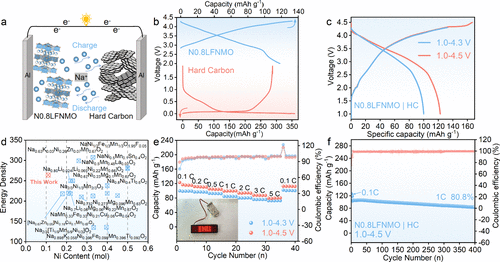
Figure 7. Electrochemical performance of the N0.8LFNMO|hard carbon (HC) full cells. (a) Schematic diagram. The first charge/discharge curves of (b) N0.8LFNMO|Na metal (blue), HC|Na metal (red) half-cells, and (c) N0.8LFNMO|HC full cell at 0.1 C. (d) Comparison of energy density with different contents of Ni in the reported full cell configurations. (e) Rate capability tests from 0.1 to 5 C. (f) Cycling performance.
总结及展望
本研究成功构建了一套高钠P2型正极材料的理性设计框架,从根本上揭示了过渡金属组分对物相形成及电化学行为的影响机制。N0.8LFNMO材料的研制不仅证明了通过高钠含量设计可以有效缓解层状氧化物的固有缺陷,更为开发低成本、高性能的钠离子电池提供了坚实的理论依据。这种从基础原理出发到实际材料设计的研发模式,突破了传统实验的随机性,为下一代高比能长寿命钠电材料的开发开辟了新的道路。未来,随着该设计准则的进一步推广,钠离子电池有望在更多高性能要求的储能场景中实现大规模应用。