【JACS】上海大学陈雨、华东理工谢贺新等|协同精准“狙击”肿瘤细胞:近红外前药助力原位癌症疫苗效能提升 68%
文章标题: Enzyme-Triggered Self-Immobilization of an NIR Prodrug for Tumor-Selective Cancer Vaccination
通讯作者: Yuyao Li, Meihua Yu, Hexin Xie, Yu Chen
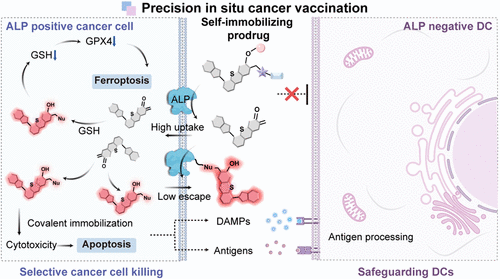
文章概要
引言
癌症免疫治疗通过激活人体自身免疫系统来识别和清除肿瘤,在临床上已取得显著成功,但肿瘤微环境的复杂异质性常导致治疗响应率参差不齐。原位癌症疫苗作为一种极具前景的策略,能够诱导垂死的肿瘤细胞释放自体抗原,从而激发特异性免疫反应。然而,目前的化疗或放疗手段往往缺乏选择性,在杀伤肿瘤的同时,也会不可避免地损伤肿瘤浸润免疫细胞,这严重削弱了免疫系统捕获抗原并启动后续免疫应答的能力。为了打破这一瓶颈,研究团队开发出一种基于酶触发自固定机制的近红外前药 ALPICD-2,旨在实现精准杀伤肿瘤细胞的同时,最大限度地保护周围的免疫“战友”,为原位癌症疫苗的临床转化提供新思路。
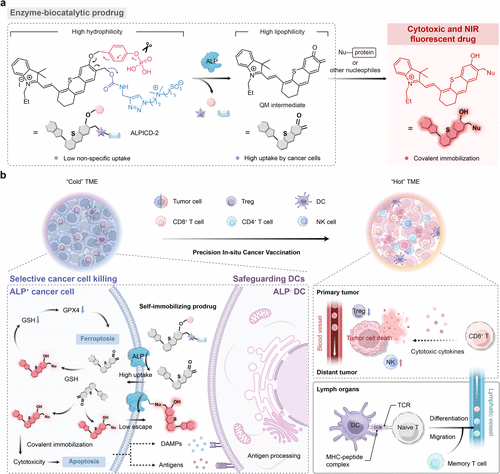
Figure 1. Schematic illustration of ALP-activatable NIR prodrug ALPICD-2 and associated immunological mechanism for cancer immunotherapy. (a) Chemical structures of ALPICD-2 and proposed ALP-promoted prodrug activation. (b) Proposed mechanism for cancer immunotherapy of ALP-activatable NIR prodrug ALPICD-2.
主要实验及结论
研究人员巧妙设计了前药分子 ALPICD-2,利用肿瘤细胞表面高度表达的碱性磷酸酶(ALP) 作为“钥匙”。实验证实,当该前药接触到肿瘤细胞膜上的 ALP 时,会发生剧烈的亲水-亲脂性转变,生成的中间体能与细胞内的蛋白质等亲核物质发生共价结合,从而实现在肿瘤细胞内部的“定向锁死”与高效富集。这一机制不仅启动了强烈的近红外荧光以便于实时追踪,更通过干扰脂质稳态诱导了肿瘤细胞发生凋亡与铁死亡的双重死亡模式。
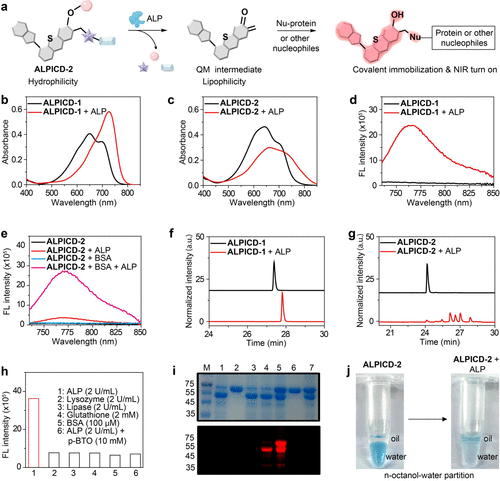
Figure 2. In vitro characterization of ALPICD-2. (a) Proposed mechanism of ALP-activatable NIR prodrug ALPICD-2. (b, c) UV–vis absorption spectra of ALPICD-1 (10 μM) or ALPICD-2 (10 μM) in PBS (pH 7.4) before and after incubation with ALP (2 U/mL) at 37 °C for 30 min. (d) Fluorescence spectra of ALPICD-1 (10 μM) before and after incubation with ALP (2 U/mL). (e) Fluorescence spectra of ALPICD-2 (10 μM) before and after incubation with ALP (2 U/mL) in the presence or absence of BSA (100 μM) in PBS at 37 °C for 30 min. (f, g) HPLC traces of ALPICD-1 (10 μM) or ALPICD-2 (10 μM) before and after incubation with ALP (2 U/mL) at 37 °C for 30 min. (h) Fluorescence response of ALPICD-2 (10 μM) to various analytes in PBS. λex/λem = 720/760 nm. (i) Coomassie blue staining (top) and fluorescent imaging (bottom) of SDS-PAGE gel. Indicated proteins were incubated with or without ALPICD-2 in PBS at 37 °C for 2 h. M: protein marker; 1: ALP; 2: BSA; 3: ALP + ALPICD-1; 4: ALP + ALPICD-2; 5: ALP + BSA + ALPICD-2; 6: BSA + ALPICD-2; 7: ALP + BSA + p-BTO + ALPICD-2. p-BTO: (−)-p-bromotetramisole oxalate, an ALP inhibitor; λex/λem = 785/820 nm. (j) Changes in the oil–water partition of ALPICD-2 (20 μM) before and after incubation with ALP (2 U/mL).
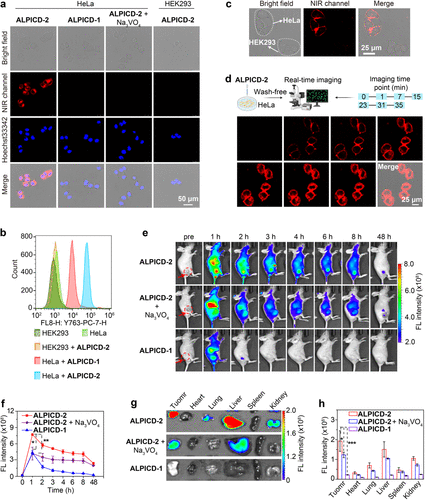
Figure 3. NIR fluorescence images of ALP in cancer cells or tumor-bearing mice with ALPICD-2. (a) Confocal NIR fluorescence images of ALP-overexpressed HeLa cells or ALP-negative HEK293 cells. ALPICD-2 or ALPICD-1 (4 μM) was incubated at 37 °C for 30 min with the indicated cells, as well as cells pretreated with Na3VO4 (10 mM), an ALP inhibitor, for 20 min before incubation with ALPICD-2. (b) Flow cytometry analysis showing the histograms of HeLa cells or HEK293 cells incubated with ALPICD-2 or ALPICD-1 (4 μM). (c) Specific labeling of ALP-overexpressed HeLa cells in the presence of ALP-negative HEK293 cells. A mixture of HeLa cells and HEK293 cells was incubated with ALICD-2 (1 μM) at 37 °C for 10 min before imaging. (d) Wash-free and real-time NIR fluorescence images of HeLa cells incubated with ALPICD-2 (1 μM). (e) Representative NIR fluorescence images of HeLa tumor-bearing mice after i.v. injection of ALPICD-2 or ALPICD-1 (0.4 μmol/kg), or together with i.t. injection of Na3VO4 (10 mM, 50 μL) before injection of ALPICD-2. Red circles point to the tumor locations in mice. (f) Time-course NIR fluorescence intensity in tumors. (g) Fluorescence images of the tumor and main organs at 48 h postinjection. (h) Fluorescence intensity of organs and tumors in (g). λex/em = 710/760 nm. Data represent mean values ± s.d. (n = 3); *P < 0.05, **P < 0.01, and ***P < 0.001.
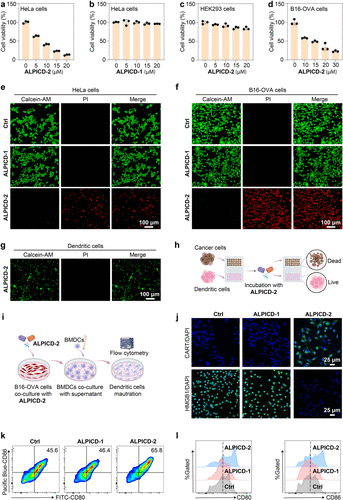
Figure 4. In vitro anticancer efficacy of ALPICD-2. (a–d) Cell viabilities of HeLa, HEK293, or B16-OVA cells upon incubation with a serial concentration of ALPICD-2 or ALPICD-1. (e–g) Live (Calcein AM)/dead (PI) staining of HeLa, B16-OVA, or DC cells incubated with ALPICD-1 or ALPICD-2. (h) Schematic illustration of ALPICD-2 selectively killing cancer cells without damaging DCs. (i) Schematic diagram of coincubating BMDCs with supernatant from ALPICD-2-treated B16-OVA cells. (j) Immunofluorescence staining images of HMGB1 and CRT levels in B16-OVA cells after diverse treatments. (k) Flow cytometry analysis showing the dot plots of CD80+CD86+ mature DCs in diverse treatment groups. (l) Representative histogram overlays of the expression of CD80 and CD86 on BMDCs in diverse treatment groups. Data represent mean values ± s.d. (n = 3).
在体外细胞实验中,ALPICD-2 展现出极高的选择性,它能精准清除 HeLa 和 B16-OVA 等 ALP 高表达的癌细胞,而对树突状细胞(DCs)等关键免疫细胞几乎无害。这种“选择性狙击”确保了肿瘤细胞死亡后释放的大量损伤相关分子模式(DAMPs)能够被完好无损的免疫细胞有效识别。动物实验结果进一步令人振奋,在 B16-OVA 荷瘤小鼠模型中,通过静脉或瘤内注射 ALPICD-2,显著增强了肿瘤组织内 CD8+ T 细胞和自然 killer (NK) 细胞的浸润与增殖。特别是在两疗程治疗后,肿瘤抑制率达到了惊人的 68%,且在双侧肿瘤模型中成功抑制了远端未处理肿瘤的生长。这表明该疗法不仅能有效清除局部病灶,还能诱发全身性的持久免疫记忆,有效防范肿瘤的转移与复发。
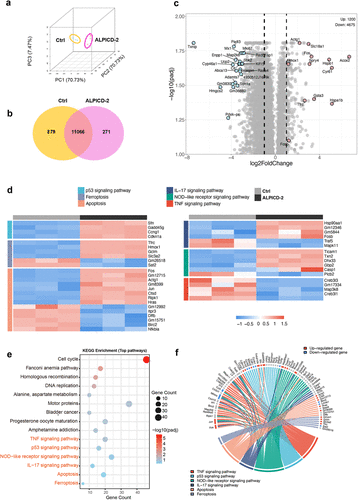
Figure 5. RNA-seq analysis associated with in vitro anticancer therapy. (a) PCA cluster analysis. (b) Venn diagram of differentially expressed genes between ALPICD-2 and control groups. (c) Volcano map of expression differences. (d) Heat map of differentially expressed genes associated with migration regulation, proliferation regulation, apoptosis, ferroptosis, and cell cycle. (e) KEGG analysis based on differentially expressed genes between ALPICD-2 and control groups. (f) KEGG path chord diagrams.
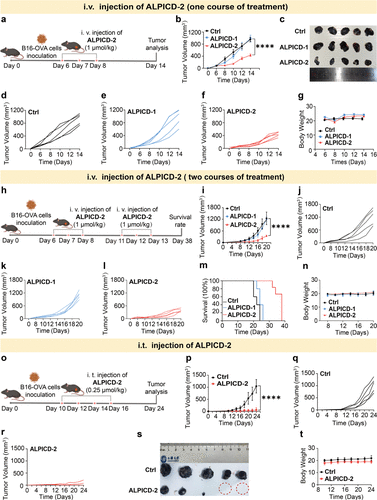
Figure 6. In vivo antitumor activities. (a) Schematic illustration of experimental schedule with one course of treatment. (b) Time-course tumor volume of mice after one course of treatment. (c) Images of dissected tumors from mice after one course of treatment. (d–f) Individual B16-OVA tumor growth volume curves of mice after one course of treatment (i.v. injection) (n = 5). (g) Time-course body weight of mice after one course of treatment. (h) Schematic illustration of experimental schedule with two courses of treatment. (i) Time-course tumor volume of mice after two courses of treatment. (j–l) Individual B16-OVA tumor growth volume curves of mice after two courses of treatment (i.v. injection) (n = 5). (m) Survival (%) of mice bearing B16-OVA tumors after two courses of treatment. (n) Time-course body weight of mice after two courses of treatment. (o) Schematic illustration of experimental schedule with i.t. injection of ALPICD-2. (p) Time-course tumor volume of mice with diverse treatment groups. (q, r) Individual tumor growth volume curves of mice with diverse treatment groups (n = 5). (s) Images of dissected tumors from mice with diverse treatment groups. (t) Time-course body weight of mice in diverse treatment groups. Data represent mean values ± s.d. (n = 3); ****P < 0.0001.
总结及展望
本研究成功构建了一个高度精准的小分子原位疫苗平台,通过“酶驱动开关”与“共价固定”策略,破解了传统疗法中抗肿瘤活性与生物安全性难以兼顾的难题。ALPICD-2 不仅是一种高效的化疗前药,更是一个强大的免疫调节器,它通过保护肿瘤微环境中的免疫“火种”,实现了从局部细胞清除到全身免疫激活的华丽转变。这一研究成果为开发具有高肿瘤累积量、低系统毒性的新一代化学免疫治疗药物提供了重要的理论依据和实践范例,未来有望在多种实体瘤的精准治疗中发挥关键作用。